Novartis Can “Fix” the Sufficiency of Its Patent 10 Years After Filing
A recent decision of the Federal Court in Novartis v Teva 2013 FC 283 (“Novartis”) [under appeal A-123-13] has established that the relevant date for patent sufficiency is not until the publication date. Novartis may highlight an inconsistency in validity analysis. In some ways the notional “person skilled in the art” (who by definition is not inventive) is expected to know more, and earlier, than the inventors themselves (who are obviously inventive).
Narrowing of claims to one compound renders patent sufficient
In Novartis, Mr. Justice Hughes held that Novartis’ compound patent (CA 1,338,937[i]) was sufficient only in 1997, after Novartis had substantially amended its patent claims (and arguably the nature/definition of its invention) since filing its patent in 1987. In fact Novartis was given the benefit of 10 years to “perfect” the description by further limiting the specific subject matter of its claimed monopoly to one compound – zoledronate. At the same time, the person skilled in the art was required to have determined in 1987 that it was “plain as day” that this one compound (as eventually selected to be claimed by the patentee) would be useful for the claimed uses.
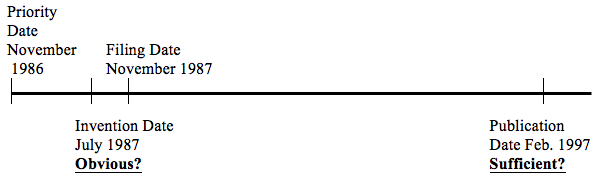
Interestingly the court first observed that “Canadian law has not been clear as to what is the date for considering sufficiency”. Hughes refers to a 2000 SCC decision that considered the relevant date of claims construction (finding it to be the publication date), however declined to consider the issue of sufficiency. The SCC stated:
While there may be some advantages to the establishment of a single critical date for multiple purposes including obviousness, sufficiency and claims construction, my view is that Canadian law does not support the date of application as the critical date for claims construction.
[Free World Trust v Électro Santé Inc, [2000] 2 SCR 1024 at para. 53]
Hughes neglected to follow UK jurisprudence establishing that sufficiency is to be evaluated at the date of filing as “An applicant should not be able to take advantage of intervening advances in the state of the art so as to render sufficient an insufficient application as filed”. Notably Hughes appears to agree with the UK approach, but finds the Teva SCC 2012 decision (regarding the Viagra patent) mandates that the publication date must be the relevant date for sufficiency:
That [UK] decision would make sense in Canada under the scheme of either the “old” or “new” Patent Act, were it not for the recent decision of the Supreme Court of Canada in Teva. [Teva v Pfizer 2012 SCC 60, “Teva SCC”]
In view of Teva SCC, the Novartis patent went from insufficient to sufficient as explained below:
| Claims | Disclosure | |
| November 1987Filing Date | – “the application contained claims to many compounds, including genus claims and claims to specific compounds; including, but not restricted to, zoledronate. If I were to consider sufficiency as of the date of filing the application, I would find that the application was no different than that considered by the Supreme Court in Teva, and thus was invalid for lack of sufficient disclosure.” | – in July 1987, the inventors had made zoledronate and tested it using a rat model accepted by the court[ii]– but the zoledronate testing is not in the patent- the disclosure refers to pages of compounds, and p. 4 refers to general tests/general ranges of “the compounds” in general
– Examples refer to 32 compounds including zoledronate that are “specifically” the invention |
| February 1997Publication (and Issue) Date | – the issued patent contains only 2 claims directed at zoledronic acid and therefore patent is sufficient | – no changes in the disclosure between filing and publication are discussed by the Court |
Applying the Teva SCC approach, and “seeking to keep up with the state of the jurisprudence”, Hughes clearly found there was a change in sufficiency between filing and publication because the claims were narrowed to one compound. As this was simply a result of legal developments (and not Novartis trying to rely on extraneous developments over 10 years[iii]), the later date should be chosen. Hughes does not specifically discuss the fact that the Teva SCC decision failed to review the proper date for sufficiency even where the claims of the Viagra patent were substantially amended by disclaimer following publication and issuance.[iv] The SCC considered the later disclaimed claims (filed 8 years after publication). Perhaps this was a moot point in Teva SCC because even after the disclaimers to “fix” its patent, Pfizer’s patent was still invalid as insufficient.
In evaluating the suitability of the publication date as the proper construction date, Hughes in fact does refer to the fact that claims may be amended following issuance, including by disclaimer. According to Hughes’ reasoning, however, it is the published claims that are to be considered for sufficiency. This approach would suggest that “disclaimed claims” cannot be used to evaluate sufficiency, as a disclaimer is only considered effective as of the filing date of the disclaimer and not before.
Hughes also takes the view that using the publication date is consistent with the fact that this is the date the public first sees the patent and “the person applying for the patent has committed to claims for the invention in a manner available to the public”. However this approach may ignore the fact that Novartis is given an additional 10 years to commit to exactly what their invention is. The person skilled in the art has to show the same invention is “plain as day” 10 years earlier. In this case Hughes found zoledronate[v] was not obvious given the “broad number of choices… there is still too much uncertainty as to whether any particular combination will be useful.” It was “largely an empirical exercise” to identify new “promising” compounds. As a result it was not “more or less self-evident” that “zoledronate … ought to work”. Of course the patentee had not even determined that one particular compound would be useful – they had tested many (and likely predicted the many pages of compounds initially claimed).
Effect on Future Patent Cases
Given the recent analysis in Teva SCC, patent claims now perhaps have greater importance in sufficiency (and not simply defining the fenced in area of the monopoly). A patentee is able to clarify the definition of his invention and how to make it, so the person skilled in the art can make it successfully, by narrowing its claims (and arguably selecting its best compounds from the thousands it may have initially started with at filing). This may in fact be a change in the specification that can certainly not be reasonably inferred from the disclosure as filed.
Novartis was able to substantially amend its claims during prosecution (and thereby “fix” its description given the significance of narrower claims in its issued patent). This is despite the fact that Patent Rule 181 says the specification cannot be amended to “describe or add matter not reasonably to be inferred from the specification … as originally filed.”
This admittedly strained analysis arises because the SCC took the rather novel approach of reviewing Pfizer’s Viagra patent by considering whether the person skilled in the art could determine what the “real compound” was that Pfizer had successfully tested and described anonymously in its patent. The SCC looked at 2 different compound claims and found the person skilled in the art would be unable to identify the “real compound”. This leads to the finding that, had there been only 1 compound claim, the person skilled in the art would not be guessing and would be able to use the invention successfully because they would know the “real compound” must be the specific claimed compound. This lead Hughes to conclude that Novartis’ patent was rendered sufficient by excluding all of the claims but claims to one single compound.
The date for sufficiency may be a rare issue – the Novartis patent was an Old Act patent (filed before 1989 and therefore only published at the time of issuance which was 10 years after filing). For “New Act” patents, filed after 1989, a patent is published just 18 months after filing/priority. It is unlikely that New Act patents would have such drastic amendments in this first 18 month period prior to publication.
Another interesting conclusion of this case is that ironically, Novartis’ patent seems to have been saved as a result of the conflict and its decision to narrow its claims to the one commercial compound (and render its patent sufficient).’
[i] http://brevets-patents.ic.gc.ca/opic-cipo/cpd/eng/patent/1338937/summary.html
[ii] The court found rats are “a good model for determining the effectiveness and potency of a candidate drug in humans” in the area of bone resorption.
[iii] In fact Novartis’ patent application was held up because of a conflict with another pending Boehringer application. Following settlement of the conflict, the patentee chose claims to zoledronate only.
[iv] CA 2,163,446 was filed and published in 1994, and issued in 1998. Two disclaimers were filed in 2002. In the Teva cases, only the lower court referred to the disclaimers and noted that all claims were restricted to treatment in men. The lower court (2009 FC 638) found the disclaimers “irrelevant” to the sufficiency/utility issues that were before it.
[v][v] In particular Hughes found the inventive concept was: “zoledronate is a useful inhibitor of bone resorption in rats and that, as such, is suitable for use as a medicament in the treatment of diseases in humans associated with impairment of calcium metabolism”.


Comments are closed.